GMP-production of engineered human myocardium for heart failure repair
- Funded period
-
2018 – 2022
- Granted budget
-
€ 2,654,615
- Indication
heart failure
- Therapeutic Principle
Tissue engineering
- Principal Investigator
-
Wolfram H. Zimmermann (University Medical Centre Göttingen)
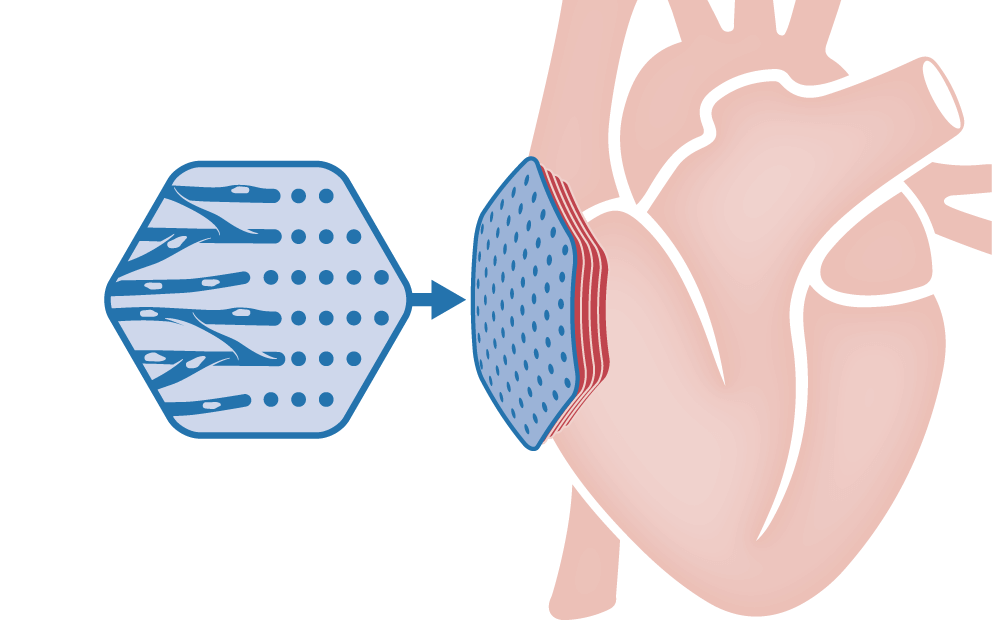
Irreversible and progressive loss of cardiomyocytes is the underlying cause of heart failure. Remuscularization of the failing heart can be achieved by epicardial implantation of tissue engineered myocardium.
A major procedural challenge for the translation of cell based therapies is the set-up and validation of a cGMP-production process. This should be done early during the translation process to reduce costs and variability/inconsistency already in preclinical studies and facilitate clinical translation into first-in patient studies. We have developed a versatile protocol for the construction of engineered human myocardium from ESCs and iPSCs to meet cGMP demands. The objectives of this project are to now set-up a GMP production pipeline and obtain a manufacturing authorization from the local competent authority for the preparation of EHM for a first in patient safety study with an anticipated number of 10 patients with end-stage heart failure
Results
The patches developed in the project were used to start the BioVAT-HF-DZHK20 clinical trial, which is funded by the DZHK clinical trials program. (press release: Start of First Clinical Trial on Tissue Engineered Heart Repair, 9.2.2021)
