Dr. Pieterjan Dierickx

Contact:
Dr. Pieterjan Dierickx
Max Planck Institute for Heart and Lung Research
Parkstraße 1
61231 Bad Nauheim, Germany
Email: pieterjan.dierickx(at)mpi-bn.mpg.de
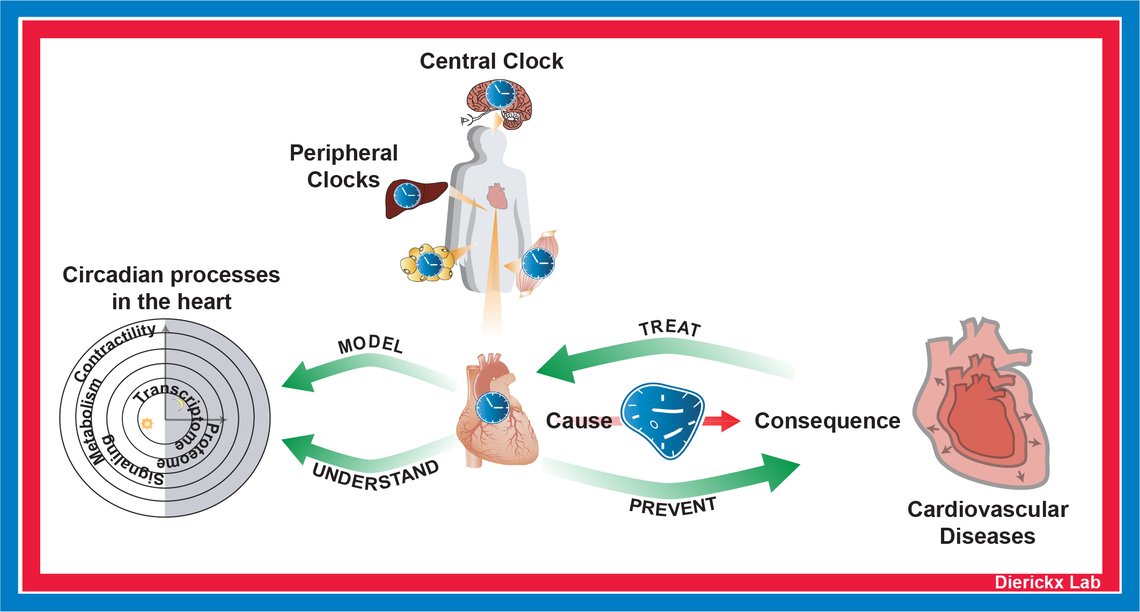
Research areas: cardiovascular health, circadian rhythms, cardiometabolism
Pieterjan Dierickx is a DZHK junior research group leader at the Max Planck Institute for Heart and Lung Research in Bad Nauheim and investigates how the internal clock influences energy metabolism in the heart. Circadian rhythms coordinate many aspects of behavior and physiology (e.g., feeding/fasting cycles, sleep to wake transitions, body temperature) to be in synchrony with the 24-hour rotation of the earth. In the heart specifically, misregulation of the circadian clock leads to metabolic defects and cardiac dysfunction. The importance of being synchronized with the environment is becoming increasingly clear, and he is convinced that there are great opportunities ahead to exploit the clock to stay healthier. In addition, a better understanding of the cardiac clock will aid the development and optimization of treatment strategies.
Research focus
The Dierickx lab is interested in how the circadian clock drives rhythmic processes in the heart. In humans, disruption of these rhythms is highly associated with increased risk for cardiovascular disease development. Importantly, clock disruption can be driven by genetic mutations, but is also severely affected by life style. The indispensability of clock proteins in the heart is mechanistically illustrated by their recent findings that loss of the circadian repressors REV-ERBa and b in cardiomyocytes leads to heart failure and premature death in mice.
Projects in the Dierickx lab are centered around the integration of next-generation sequencing techniques, whole-body physiology analysis in specialized genetic mutant mouse lines and the use of cardiac in vitro models. Pieterjan Dierickx his team aims to develop treatment strategies for cardiometabolic insufficiency and will use data from human patients to better understand how deregulation of the clock contributes to heart disease. Investigating how the circadian clock and its intricate connection to rhythmic metabolic programs can be deployed to tackle prevention, diagnosis and treatment of heart failure will be the overarching goal.
Major achievements and awards
- Deutsche Gesellschaft für Kardiologie (DGK, German Cardiac Society): Science Prize of the AG 23 Heart and Diabetes (2023)
- German Centre for Cardiovascular Research (DZHK): Junior Research Group Grant (2022)
- American Heart Association (AHA): Postdoctoral Fellowship (2019)
- Netherlands Heart Institute (NHI): Postdoctoral Fellowship (2017)
- Fonds voor Wetenschappelijk Onderzoek (FWO): PhD Fellowship (2011)
- The company of Biologist: Travel Grant (2022)
Key publications
Carpenter BJ, Lecacheur M, Mangold YN, Cui K, Günther S, Pieterjan Dierickx**. NAD+ controls circadian rhythmicity during cardiac aging. bioRxiv (2023). doi: https://doi.org 10.1101/2023.11.02.565150
Latimer MN, Williams LJ, Shanmugan G, Carpenter BJ, Lazar MA, Dierickx P, Young ME. Cardiomyocyte-specific disruption of the circadian BMAL1-REV-ERBα/β regulatory network impacts distinct miRNA species in the murine heart. Communications Biology (2023). PMID: 37952007
Carpenter BJ and Dierickx P**. Circadian NAD+ metabolism, from transcriptional regulation to healthy aging. American Journal of Phyisology-Cell Physology (2022). PMID: 36062878
Dierickx P**, Carpenter BJ, Celwyn I, Kelly DP, Baur JA, and Lazar MA#. Nicotinamide riboside Improves cardiac function and prolongs survival after disruption of the cardiomyocyte clock.
Frontiers in Molecular Medicine (2022). DOI: 10.3389/fmmed.2022.887733
Dierickx P**, Zhu K, Carpenter BJ, Jiang C, Vermunt MW, Xiao Y, Luongo TS, Yamamoto T, Martí-Pàmies Í, Mia S, Latimer M, Diwan A, Zhao J, Hauck AK, Krusen B, Nguyen HCB, Blobel GA, Kelly DP, Pei L, Baur JA, Young ME, and Lazar MA**. Circadian REV-ERBs repress E4bp4 to activate NAMPT-dependent NAD+ biosynthesis and sustain cardiac function. Nature Cardiovascular Research (2021). PMID: 35036997
(Highlighted in Circulation)
Chirico N, van Laake LW, Sluijter JPG, van Mil A, and Dierickx P**. Cardiac circadian rhythms in time and space: The future is in 4D. Current Opinion in Pharmacology (2020). PMID: 33338891
Guan D, Xiong Y, Trinh TM, Xiao Y, Hu W, Jiang C, Dierickx P, Jang C, Rabinowitz JD, Lazar MA. The hepatocyte clock and feeding control chronophysiology of multiple liver cell types. Science (2020). PMID: 32732282
Dierickx P, Emmett MJ, Jiang C, Uehara K, Liu M, Adlanmerini M, and Lazar MA. SR9009 has REV-ERB-independent effects on cell proliferation and metabolism. PNAS (2019). PMID: 31127047
Dierickx P**, van Laake LW. & Geijsen N. Circadian clocks: from stem cells to tissue homeostasis and regeneration. EMBO Reports (2017). PMID: 29258993
du Pré BC*, Dierickx, P*, Crnko S, Doevendans PA, Vos MA, Geijsen N, Neutel D, van Veen TAB, and van Laake LW. Neonatal rat cardiomyocytes as an in vitro model for circadian rhythms in the heart. Journal of Molecular and Cellular Cardiology (2017). PMID: 28823816
Dierickx P**, Vermunt MW, Muraro MJ, Creyghton MP, Doevendans PA, van Oudenaarden A, Geijsen N, and van Laake LW#. Circadian networks in human embryonic stem cell-derived cardiomyocytes. EMBO Reports (2017). PMID: 28536247
Tiburcy M, Hudson JE, Balfanz P, Schlick S, Meyer T, Chang Liao ML, Levent E, Raad F, Zeidler S, Wingender E, Riegler J, Wang M, Gold JD, Kehat I, Wettwer E, Ravens U, Dierickx P, van Laake LW, Goumans MJ, Khadjeh S, Toischer K, Hasenfuss G, Couture LA, Unger A, Linke WA, Araki T, Neel B, Keller G, Gepstein L, Wu JC, and Zimmermann WH. Defined Engineered Human Myocardium with Advanced Maturation for Applications in Heart Failure Modelling and Repair. Circulation (2017). PMID: 28167635
*These authors contributed equally to this work
** Corresponding author
