Dr. Nuno Guimarães-Camboa

Contact:
Dr. Nuno Guimarães-Camboa
Goethe University
Institute for Cardiovascular Regeneration
Theodor-Stern-Kai 7
69590 Frankfurt am Main
Email: ncamboa@med.uni-frankfurt.de
Research areas: vascular biology, aneurysmal diseases, cardiac fibrosis
Dr. Nuno Guimarães-Camboa is DZHK Junior Research Group Leader at the Goethe University in Frankfurt. He studies transcriptional mechanisms operating in perivascular cells in homeostasis and disease
Research focus
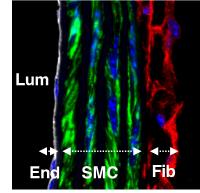
Dr. Camboa’s group studies the roles of perivascular cells in homeostasis and disease. Our bodies have a complex network of blood vessels bringing oxygen and nutrients to all organs. The wall of these blood vessels is composed of different cell types organized in concentric rings. Endothelial cells delimitate the vascular lumen and are externally surrounded by perivascular cells (pericytes, vascular smooth muscle, adventitial fibroblasts). Perivascular cells are required for vascular stability and play a central role in aneurysmal diseases. In addition, due to their capacity to regulate vascular permeability and potential to function as tissue-resident stem cells, perivascular cells also play critical roles in diseases as diverse as cancer, obesity, and organ fibrosis. The group of Dr. Camboa uses a combination of in vitro and in vivo models to study gene expression mechanisms controlling the properties of perivascular cells in physiological and pathological settings.
Dr. Camboa hopes that, in the long term, it will be possible to use our understanding of the roles of perivascular cells and molecular pathways controlling their behavior to develop novel therapeutic and/or diagnostic strategies for patients suffering from aneurysms or cardiac fibrosis.
Major achievements and awards
- 2018 – Junior Research Grant from the DZHK Excellence Program
- 2016 – Schulman Award for Outstanding Research. University of California, San Diego (USA)
Key Publications
Cattaneo P*, Mukherjee D*, Spinozzi S, Zhang L, Larcher V, Stallcup WB, Kataoka H, Chen J, Dimmeler S, Evans SM and Guimarães-Camboa N. (2020) Parallel lineage-tracing studies establish fibroblasts as the prevailing in vivo adipocyte progenitor. Cell Reports; 30(2): 571–582.
Moore-Morris T.*, Cattaneo P.*, Guimarães-Camboa N., Cedenilla M., Banerjee I., Ricote M., Kisseleva T., Brenner D.A., Gu Y., Dalton N.D., Peterson K.L., Chen J., Pucéat M., Evans S.M. (2017). Infarct fibroblasts do not derive from bone marrow lineages. Circulation Research, 122(4):583-590.
Guimarães-Camboa N., Cattaneo P., Sun Y., Moore-Morris T., Gu Y., Dalton D., Rockenstein E., Masliah E., Peterson K., Stallcup W.B., Adams R., Chen J., Evans S.M. (2017). Pericytes of Multiple Organs Do Not Behave as Mesenchymal Stem Cells In Vivo. Cell Stem Cell, 20, 345–359.
Guimarães-Camboa N., Stowe J., Aneas I., Sakabe N., Cattaneo P., Henderson L., Kilberg M.S., Johnson R.S., Chen J., McCulloch A.D., Nobrega M.A., Evans S.M., Zambon A.C. (2015) HIF1α Represses Cell Stress Pathways to Allow Proliferation of Hypoxic Fetal Cardiomyocytes. Developmental Cell, 33: 507-521.
Moore-Morris T., Guimarães-Camboa N., Banerjee I., Zambon A.C., Kisseleva T., Velayoudon A., Stallcup W.B., Gu Y., Dalton N.D., Cedenilla M., Gomez-Amaro R., Zhou B., Brenner D.A., Peterson K.L., Chen J., Evans S.M. (2104) Resident fibroblast lineages mediate pressure overload–induced cardiac fibrosis. JCI 124(7):2921-2934.
